 In a steam distribution system the pressure is always more than 0 bar gauge.Īt 7 bar g ( absolute 8 bar) the saturation temperature of water is 170.42 oC. 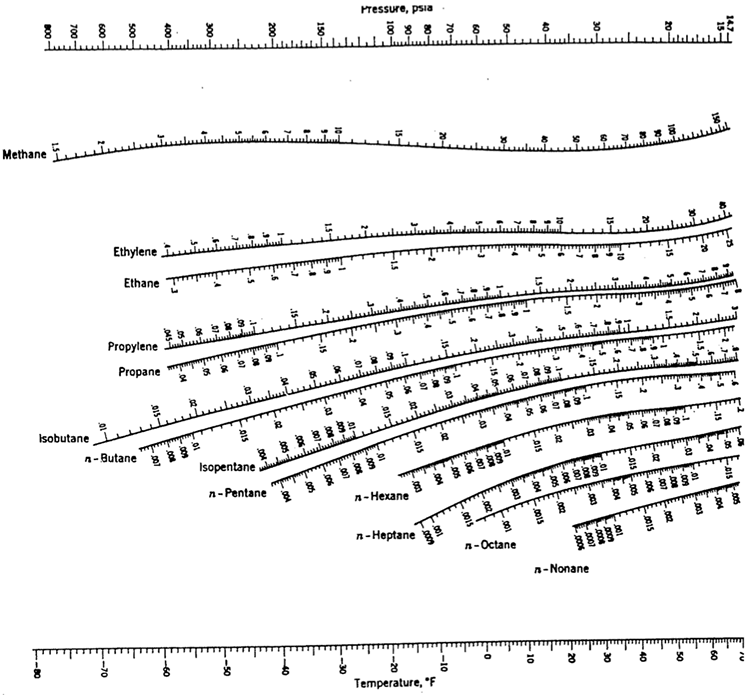
Steam at atmospheric pressure is of a limited practical use because it cannot be conveyed by its own pressure along a steam pipe to the points of use. = 2675.43 kJ/kg Example - Boiling Water at 170 oC and 7 bar (700 kPa) Atmospheric Pressure The total specific enthalpy for steam at 0 bar gauge is: Therefore at 0 bar g ( absolute 1 bar) the specific enthalpy of vaporation is 2257.19 kJ/kg. Therefore the specific enthalpy of water at 0 bar g (absolute 1 bar ) and 100 oC is 417.51 kJ/kg.Īnother 2257.92 kJ of energy is required to evaporate 1 kg of water at 100 oC into 1 kg of steam at 100 oC.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
